The U.S. National Institutes of Health (NIH) held its Joint Martin Delaney Collaboratories Towards an HIV-1 Cure meeting on December 14 – 15, 2021. The purpose of the meeting was to introduce the newly funded Collaboratories to each other and to the public. In August 2021, the NIH expanded the Martin Delaney program by 75% to ten Collaboratories to advance the search towards an HIV cure.

Martin Delaney in 1997.
The Delaney Collaboratories are named after HIV/AIDS treatment activist Martin (“Marty”) Delaney and represent the flagship NIH program on HIV cure research, focusing on basic science, which will increase the knowledge base of the HIV cure field, and applied science, which will use that knowledge to solve specific problems. The Martin Delaney program aims to foster collaboration between academic, industry, government, and community partners towards finding an HIV cure or durable antiretroviral (ART)-free HIV suppression.
The first iteration (2011 – 2015) of the Martin Delaney Collaboratories included 3 Collaboratories ($US 70 million). The second iteration (2016 – 2020) counted 6 Collaboratories ($154 million). The third iteration (2021 – 2025) now includes 10 Collaboratories and will allocate approximately $269 million towards an HIV cure research in the next 5 years. This work is important as it will accelerate the pace of HIV cure research and community, patient, and stakeholder engagement towards an HIV cure.
The community looks forward to new HIV cure research discoveries in the years to come, and is encouraged by this increased investment from the NIH towards finding an HIV cure.
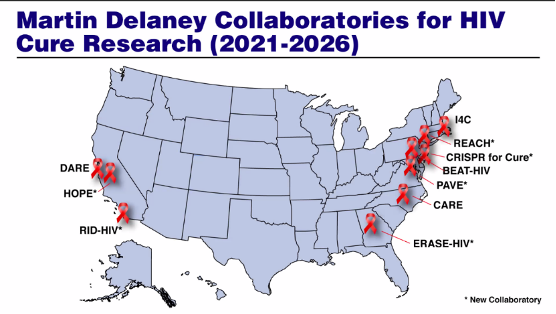
The 10 newly-funded (or renewed) Martin Delaney Collaboratories include:
| DARE Collaboratory v3.0 headquartered at the University of California San Francisco, under the leadership of Dr. Steven Deeks. The goal of DARE v3.0 is to develop strategies that enhance the ability of the immune system to control HIV in the absence of therapy and eliminate it completely. | |
| CARE Collaboratory v3.0 headquartered at UNC Chapel Hill, under the leadership of Dr. David Margolis. The goal of CARE v3.0 is to advance HIV latency reversal (or reawakening HIV reservoirs – which is the main barrier to HIV cure) and clearance of virus. | |
| BEAT-HIV v2.0 headquartered at the Wistar Institute in Philadelphia, under the leadership of Dr. Luis Montaner. The goal of BEAT-HIV v2.0 is to develop strategies to reduce the size of the HIV reservoir and achieve durable control of HIV off ART. | |
| I4C Collaboratory v2.0 headquartered at the Beth Israel Deaconess Medical Center (Harvard Medical School) in Boston, under the leadership of Dr. Dan Barouch. The goal of I4C v2.0 is to advance scientific understanding of viral reservoir and develop immunologic strategies for HIV-1 remission, such as broadly-neutralizing antibodies. | |
| REACH Collaboratory v1.0 headquartered at Weill Cornell Medicine in New York City, under the leadership of R. Brad Jones and co-led by Dr. Marina Caskey of Rockefeller University. The goal of REACH v1.0 is to harness cellular immune responses to achieve durable remission and elimination of HIV reservoirs. | |
| HOPE Collaboratory v1.0 headquartered at the Gladstone Institute, in San Francisco, under the leadership of Dr. Melanie Ott. The goal of HOPE v1.0 is to use epigenomic (what regulates expression of genes) and genomic (or gene-based) approaches to permanently inactivate HIV. | |
| RID-HIV Collaboratory v1.0 headquartered at Scripps Research in San Diego, under the leadership of Dr. Sumit Chanda, and co-led by Drs. Paula Cannon (University of South California) and Rafick Sékaly (Emory University). RID-HIV is focused on the discovery and validation of interventions to cure HIV by testing innovative concepts. | |
| CRISPR for Cure Collaboratory v1.0 headquartered at Temple University, in Philadelphia, under the leadership of Dr. Kamel Kalili. The goal of CRISPR for Cure v1.0 is to advance CRISPR-based (gene) therapies as a curative approach to HIV infection. | |
| ERASE-HIV Collaboratory v1.0 headquartered at Emory University, in Atlanta, under the leadership of Drs. Deanna Kulpa, Mirko Paiardini and Guido Silvestri. The goal of ERASE-HIV v1.0 is to develop innovative immunological approaches. | |
| PAVE Collaboratory v1.0 headquartered at Johns Hopkins University, in Baltimore, under the leadership of Dr. Deborah Persaud and co-led by Dr. Ann Chahroudi from Children’s Healthcare of Atlanta. The goal of PAVE v1.0 is to accelerate search for a cure for HIV in children and adolescents. |
|
Principal Investigator(s)
|
Scientific Approach
|
Industry Partner(s)
|
Community Partner(s)
|
Website
|
|
|
DARE v3.0 (Delaney AIDS Research Enterprise) |
|
Immune-based therapies and cell and gene therapies |
|
||
|
CARE v3.0 (Collaboratory of AIDS Researchers for Eradication) |
David M. Margolis, UNC Chapel Hill |
Reactivation of HIV reservoirs and clearance of virus |
|
||
|
BEAT-HIV v2.0 |
|
Strategies for durable control of rebound |
|
||
|
I4C v2.0 |
|
Immunotherapy for HIV cure |
Los Alamos National Laboratory |
||
|
REACH v1.0 (Research Enterprise to Advance a Cure for HIV) |
|
Cellular immune responses (natural killer and T cells) to achieve durable remission of HIV |
|
Forthcoming |
|
|
HOPE v1.0 (HIV Obstruction by Programmed Epigenetics) |
|
‘Block, lock and excise’:
|
Amgen |
Community Arts Integrated Research |
|
|
RID-HIV v1.0 (Reversing Immune Dysfunction for HIV Eradication) |
|
Reversing immune dysfunction through various innovative approaches |
Merck |
||
|
CRISPR for Cure v1.0 |
|
CRISPR-based (gene) therapies |
Excision Biotherapeutics |
||
|
ERASE-HIV v1.0 (Enterprise for Research & Advocacy to Stop & Eradicate HIV) |
|
Innovative immunological approaches to:
|
|
||
|
PAVE v1.0 (Pediatric Adolescent Virus Elimination) |
|
Accelerate search for a cure for HIV in children and adolescents |
Forthcoming |
For more information: